|
|
Introduction
Initial attempts to grow skin and its cellular components centered on the use of organ cultures and
explants cultures, where whole pieces of skin were kept alive and growth was confined to the epipole
around the piece or onto the plastic around the explants. These cultures have a short life span and
limited applications, as mixed cultures of keratinocytes, melanocytes, Langerhans cells, fibroblasts,
Merkel’s cells, nerve cells and glandular cells are obtained [1].
To understand the function and dysfunction of this complex structure,
it is necessary to study partial aspects in models of manageable size and to reintegrate the
results back into context, i.e., establishment of pure cell culture of each cellular component of
the skin. During the past three decades selective cultivation of cell components of skin has been
achieved and considerably added to our understanding of their biology in health and disease.
Furthermore, cultivated cells could be reassembled according to experimental design and indications
of their use.
Pure cell culture and reconstructed human skin equivalents as an alternative to animal
experimentation offer, not only a way to concede to demands of regulatory authorities,
animal welfare organizations, consumers and scientists, but also provide a means to improve and
extend our knowledge of biological processes in the skin. Molecular definition of disease at
the level of the gene and advances in recombinant DNA technology suggest that many diseases are
amenable to correction by genes not bearing the defective elements that result in disease.
By the use of cell cultures dermatopharmacology has also been enormously stimulated. In addition,
modern molecular genetics has been utilized to distinguish between benign and malignant cells
providing a mean of targeting the abnormal mutant gene by very specific and precise therapeutic
modality, e.g., gene and antisense therapy [2]. In this regard, tissue culture
models are ranging from simple monolayer cultures of one cell type to co-cultures of epithelial and
mesenchymal cells and finally to three dimensional skin equivalents-organotypic cultures-that form
a highly organized epithelium under the control of co-cultured fibroblasts [3].
In skin equivalents, epidermal keratinocytes grow exposed to air on a matrix of type I collagen
in which either primary dermal fibroblasts or heterologous mouse fibroblast lines and possibly
other cells are embedded, giving rise to almost normal tissue architecture [4].
Presently, various skin reconstructs are available composed either of the epidermal compartment
only or of both the epidermal and dermal compartments. Within each compartment various types of
cells can be incorporated, including keratinocytes, melanocytes and Langerhans cells in the
epidermal, and fibroblasts and endothelial cells in the dermal compartment. The quality of the
human skin equivalents has reached a point that their suitability for skin toxicity testing will
make great progress. Next to the field of toxicity and safety standards, skin equivalents offer
a well-characterized model for studies of the basic skin biology, wound repair, regulation of
melanogenesis, pathogenesis of skin diseases and skin cancer [5].
Melanocyte Culture
Melanocytes are highly differentiated neural crest
derived cells responsible for synthesizing melanin which is
transferred in the melanosomes through dendritic processes
to surrounding epidermal keratinocytes. The problem of
overgrowth of other epidermal and dermal cells, such as
keratinocytes and fibroblasts in culture has been solved and
in the past few decades, selective cultivation of human
melanocytes from newborn and adult skin has been achieved
[6].
Tetradecanoyl phorbol acetate (TPA), cholera toxin and serum
containing Dulbecco’s minimal essential medium (DMEM) was
the first described melanocyte growth medium. Obtained
melanocytes appear slender with a central bulge containing
their vesicular nuclei. On both poles, small globular
swellings are always observed that represent melanosomes
[7]. It has been shown that melanosomes are phagocytosed by
surrounding keratinocytes, a process that is controlled by
keratinocyte related protease-activated receptor-2 [8]8.
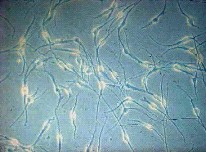
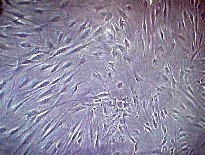
Newborn melanocytes usually display two polar dendrites,
whereas adult melanocytes frequently have several branched
dendrites (Fig. 1) [7].
Because of the presence of serum, melanocytes grown under
these conditions are not optimal for examination of factors
that may influence melanocyte function as serum may contain
substances having functional similarities with those being
tested and hence their effects could be masked. Also, the
presence of TPA, a potential carcinogenic agent, renders
melanocytes grown under these conditions not suitable for
therapeutic purposes. Nevertheless, this medium has also
several advantages. For example, pure melanocyte cultures
are obtained within a few days due to the toxic effects of
phorbol esters on keratinocytes. Furthermore, melanocytes
can be passaged up to 12-14 passages, enabling
reproducibility of the experimental trials, thus posing
additional validity to the results obtained in these
experiments [7]. In order to circumvent these drawbacks, a
serum free and hormone supplemented medium has been
successfully used for melanocyte cultivation. The used
medium (MCDB 153) supports growth of both keratinocytes and
melanocytes, but it is not optimal for growth of
fibroblasts. Crude bovine pituitary extract (BPE), cholera
toxin, and basic fibroblast growth factor (bFGF) were found
essential for melanocyte proliferation. Addition of fetal
calf serum (2%) to cultures at time of inoculation and with
every passage enhances attachment of the cultivated cells to
plates and stimulates their growth. Serum is essential for
attachment of adult melanocytes, but not for those of
newborn origin, though, melanocyte yield in the absence of
serum is low. Keratinocytes and melanocytes grow usually
together, however, after 3-4 weeks in adult cultures and 5-7
weeks in newborn cultures the keratinocytes differentiate
and separate; whereas, the melanocytes continue to grow and
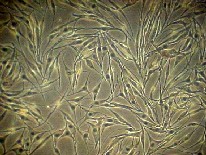
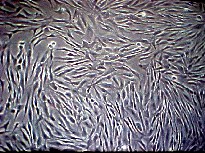
pure melanocyte cultures are obtained (Fig. 2).
 |
 |
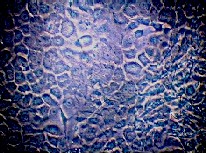
| Fig. 1: Melanocytes grown in TPA and serum. Cells are slender showing dendrites with terminal swellings |
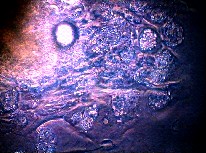
Fig. 2: Melanocytes grown in a serum free medium showing fibroblast-like morphology |
Newborn melanocytes were found to be bipolar, slender and
tended to be less dendritic in comparison with those of
adults; both have a fibroblast-like morphology. Moreover,
melanocytes obtained from newborn with dark skin showed
strong proliferative capacity, were more dendritic and
contained more melanin. Newborn melanocytes could be
passaged up to 5-6 passages, whereas those of adults up to
3-4 passages only. The identity of the cultured melanocytes
is confirmed by being DOPA+, S.100+, HMB 45+ and K.1.2.58+
and by being negative for the other epithelial cell markers,
such as CK1, CK10 for keratinocytes and CD1a for Langerhans
cells [6, 9].
Melanocytes were almost always seen in contact with the
adjacent keratinocyte colonies with dendritic projections
and bridged space between colonies. Interestingly, addition
of viable keratinocytes to melanocyte cultures showing
senescence resulted in restoration of their viability and
proliferative activity. It has also been shown that
keratinocyte conditioned medium induced melanocyte
proliferation and melanization. These observations indicate
that keratinocytes synthesize and release factors that
modulate melanocyte function. Even in vitro, a close
anatomical and functional relationship between melanocytes
and keratinocytes exists, thus posing additional validity on
the concept of epidermal melanin unit [10]. Cultivation of
nevus cells confirmed its melanocytic origin and that the
morphologic and behavioral differences observed between
epidermal melanocytes and nevus cells in the skin are the
result of local environmental influences rather than
intrinsic cellular differences [11].
Other factors that have been found to induce melanocyte
proliferation are α-MSH, endothelins, leukotrienes,
prostaglandins and several others released from adjacent
keratinocytes and fibroblasts. In addition, ultraviolet
light was found to induce melanocyte melanization and a
slight inhibition of melanocyte proliferation. Ultraviolet A
(UVA) irradiation did not substantially affect melanocytes
whereas ultraviolet B (UVB) irradiation directly activated
cultivated melanocytes. On the other hand, IL-1α, IL-6, TNF-α
and interferon ß inhibit both melanocyte proliferation and
melanin synthesis. These cytokines are released from
epidermal and dermal cells in several inflammatory skin
conditions and it has been suggested that postinflammatory
hypo- and/or hyperpigmentation may result from the effect of
these factors on epidermal melanocytes and or epidermal
melanin unit. The epidermal melanin unit has now been
extended to include not only melanocytes and neighboring
keratinocytes, but also Langerhans cells, trafficking
lymphocytes and dermal fibroblasts (Table. 1)
[12-14].
Table (1): Growth factors and their characteristics
[12-14].
|
Growth factor
|
Site
|
Biologic effect
|
Family
|
|
EGF
|
Almost all body fluids, platelets
|
Mitogenic for most epithelial tissues, fibroblasts, endothelial cells.
|
The EGF family includes EGF, TGF-α, VEGF and HBEGF.
|
|
TGF-α
|
DC, eosinophils
|
Similar to EGF, but more potent angiogenesis factor
|
|
|
HBEGF
|
DC
|
Mitogenic for keratinocytes.
|
|
|
TGF-β
|
DC, lymphocytes, fibroblasts, keratinocytes, platelets
|
Inhibits replication of most cells in vitro (keratinocytes, endothelial cells, lymphocytes, macrophages) may inhibit or stimulate fibroblasts.
|
TGF-β family (TGF-β1-5) Only TGF-β1-3 are found in mammalian cells and have similar effect.
|
|
IGF-1
|
Most tissues, fibroblasts, DC
|
Mitogenic for fibroblasts, bone cells, neural tissues, haematopoietic cells, endothelial cells.
|
The IGF-1 family (IGF-I and IGF-II). Both have similar structures and effects.
|
|
PDGF
|
Endothelial cells, platelets, DC, fibroblasts
|
Mitogenic for vascular smooth muscles, fibroblasts.
|
The PDGF family consists of PDGF and VEGF
|
|
VEGF
|
Pituitary cells
|
Mitogenic for endothelial cells but not keratinocytes, smooth muscles or fibroblasts.
|
|
|
FGF
(acidic and basic)
|
Fibroblasts, astrocytes, endothelial cells, smooth muscles
|
Mesenchymal and neural tissue mitogen
|
The FGF family (aFGF, bFGF, FGF-5, FGF-6). Only the first 3 have been extensively studied
|
|
KGF
|
Fibroblasts
|
Mitogenic for epithelial cells but not fibroblasts or endothelial cells
|
|
|
DC = dendritic cells, EGF= epidermal growth factor, FGF= fibroblast growth factor, HBEGF= heparin-binding EGF, IGF= insulin growth factor, KGF= keratinocyte growth factor, PDGF= platelet-derived growth factor, TGF= transforming growth factor, VEGF= vascular endothelial growth factor.
|
By the year 2004, melanogenic paracrine and autocrine
networks have been elucidated between melanocytes and other
types of skin cells. These include endothelin (ET)-1,
granulocyte macrophage colony stimulating factor (GM-CSF),
membrane type stem cell factor (SCF) and growth-related oncogene-α (GROα) for interactions between keratinocytes and
melanocytes, and hepatocyte growth factor (HGF) and soluble
type SCF for interactions between fibroblasts and
melanocytes [15]. These networks are also associated with
corresponding receptors expressed on melanocytes, including
ET B receptor and the SCF receptor, c-KIT. Consistent with
in vitro findings on the melanogenic paracrine or autocrine
cytokine networks, it has been found that the up- or
down-regulation of such networks is intrinsically involved
in vivo in the stimulation of melanocyte functions in
several epidermal hyper- or hypo-pigmentary disorders. These
are ET- 1/ET B receptor as well as membrane type SCF/c-KIT
for ultraviolet B-melanosis, GM-CSF for ultraviolet A-melanosis,
ET-1/ET B receptor as well as membrane type SCF for lentigo
senilis, GROα for Riehl’s melanosis,
sphingosylphosphorylcholine for hyperpigmentation in atopic
dermatitis, ET-1 for seborrheic keratosis, soluble type SCF
as well as HGF for dermatofibroma and cafe´ -au-lait macules,
and c-KIT for vitiligo vulgaris. These unveiled regulatory
mechanisms involved in the abnormal increased or decreased
levels of lesional melanocyte function provide new insights
into therapeutic tools utilizing blockage of responsible
cytokine networks [16].
Cultivated melanocytes have also enriched our knowledge
about the pathogenesis of several diseases. Sera of active
vitiligo patients exerted cytotoxic effect directly and via
the classical pathway of complement activation on
melanocytes in vitro, a finding that has been refuted by
others. Immunofluorescence studies using living cultivated
melanocytes as a substrate revealed deposition of
immunoglobulins not only on surface of cultivated
melanocytes but also on cultivated keratinocytes and
fibroblasts. At the same time, an antibody dependent
cellular cytotoxicity (ADCC) and specific melanocyte-specific
cytotoxic T cells have been detected. This observation
further supports the involvement of the immune system in
melanocyte damage in vitiligo [16]. On the other hand, sera
of alopecia areata failed to exert any cytotoxic effect on
cultured melanocytes casting doubt on involvement of these
cells in the pathogenesis of alopecia areata [18]. Western
blotting analysis using antigens obtained from cultivated
melanocytes in both diseases was also inconclusive [16].
The effect of various agents and drugs has also been
examined on cultured melanocytes. It has been shown that
nitric oxide (NO), released by inflammatory cells and
Langerhans cells is toxic to melanocytes which may play a
role in disorders associated with loss of pigmentation [19].
Retinoids reduce tyrosinase activity and therefore are used
to regulate pigmentation and lighten hyperpigmented skin.
All-trans-retinoic acid (tretinoin) enhanced the toxic
effect of hydroquinone on melanocytes [20]. Other inhibitors
of melanogenesis by competitive inhibition of tyrosinase are
arbutin, kojic acid and possibly niacinamide [21].
Ultraviolet light induced melanocyte cytotoxicity but in
vivo it results in melanocyte proliferation and melanization
which are due to a paracrine effect. Sera obtained from
psoralen plus ultraviolet A (PUVA) responding vitiligo
patients induced a significant melanocyte and dermal
fibroblasts proliferation indicating that this therapy may
have a systemic effect [22]. Reconstructed melanocyte
bearing skin is currently used in evaluation of sun
screening agents [23].
Normal human melanocytes provided the proper control of
melanoma cell research. IFN-α inhibited melanoma cell
proliferation in vitro and it is currently an established
adjuvant therapy for advanced cases of melanoma. In
addition, several oncogenes, e.g., B-raf gene, and adhesion
molecules, e.g., E- and N-cadherins, are currently evaluated
for being potential targets for therapy and prevention of
melanoma progression [24].
Because of the absence of serum and tumor promoting factors,
the serum free and hormone supplemented medium could be
suitable not only for investigative but also for therapeutic
purposes, such as transplantation procedures in patients
with vitiligo, piebaldism and other depigmentary disorders.
Indeed, several trials have been carried out with successful
results and melanocyte transplantation has become one of the
established lines of treatment of several recalcitrant
hypopigmentary and depigmentary disorders. In the
transplantation procedure, either pure melanocytes or
epidermal sheet- bearing melanocytes could be utilized [25].
Keratinocyte Culture
Keratinocytes have been successfully cultivated in 1975
when Rheinwald and Green reported the ability to grow pure
keratinocytes using a serum containing DMEM/Ham’s F12 medium
and a feeder layer (irradiated well-defined transfected
mouse 3T3 cells). Shortly thereafter the culture cell yield
has been improved by the incorporation of keratinocyte
mitogens, such as epidermal growth factor (EGF) and cAMP-elevating
agent, the cholera toxin [26].
In cultures, keratinocytes attach as single cells or small
clusters and then grow at the periphery of the colony whilst
stratifying in the center. The cells form intercellular
desmosomes and so grow as coherent colonies until confluent
stratifying multilayered sheets are obtained. These have
poorly formed squames and do not show normal skin
morphology, with cells being flattened, attenuated and
forming no stratum granulosum or stratum corneum. Membrane
coating granules and keratohyaline granules are sparse and
the cultures show the same phenotype as regenerating
epidermis [27].
Since the fundamental breakthrough of keratinocyte culture,
other ways trying to avoid serum and mouse transfected
feeder layer (3T3 cells) in growing keratinocytes have been
developed. The search for a defined serum-free medium has
resulted in a low–calcium MCDB 153 medium containing several
growth factors, which is now commercially available. In low
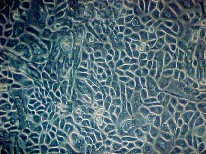
calcium medium (<0.06 mM), the cells fail to form desmosomal
interconnections and are spaced out as monolayer (Fig. 3).
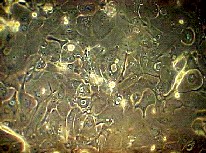
Although the keratinocytes fail to stratify, they commence
terminal differentiation with the expression of involucrin
in the larger cells which move suprabasally when calcium
levels are restored (to 1.2 mM) (Fig. 4) [28]. The cell
cycle is around 22 h with a growth fraction of 60-70%. These
systems are therefore heavily weighted towards
hyperproliferation [29]. In low Ca++ medium keratinocytes
expressed cytokeratins of germinative basal epidermis,
namely CK 5 and 14, whereas in high Ca++ medium they
expressed cytokeratins of differentiated epidermis, namely
CK 1 and 10. BPE was an essential mitogen for keratinocyte
proliferation and cells obtained from old individuals showed
weak proliferative capacity that hampered their use in
autografting [30]. In contrast to epidermal keratinocytes,
those obtained from the outer root sheath of hair follicles
have extensive proliferative capacity irrespective of donor
age, which probably depends on the residing pluripotent
epithelial stem cells. Interestingly, autologous outer root
sheath keratinocytes are successfully used in grafting of
recalcitrant chronic leg ulcers. The use of autologous
keratinocytes for grafting depends on mechanical stability
of the graft that can be improved in hybrid models.
Hyaluronic acid membranes, also known as keratinocyte
delivery system, have been proved to be extremely helpful
and textile spacers are under investigation for scaffolding
cultivated skin structures with plasticity and ease of
degradability [31].
|
|
| Fig. 3: Keratinocytes grown under low Ca++ level. Cells appear as polygonal and monolayer with prominent nuclei. |
Fig. 4: Keratinocytes grown under high Ca++ level. Cells stratify and show evidence of differentiation. |
In skin equivalents, keratinocytes tend to form a more
differentiated epidermis, with stratum corneum and
granulosum including keratohyaline and membrane coating
granules. Although these complex cultures are useful for
pharmacological experiments, they provide little tissue
expansion, whereas in monocellular cultures one cm of skin
can generate 1 m culture area within 6 weeks. This huge
population expansion has permitted the use of keratinocyte
cultures for skin grafting and keratinocyte grafting. This
has been applied to burns, difficult to heal chronic leg
ulcers, excision sites of giant melanocytic nevi and
correction of hypospadia [32-34].
Both autologous keratinocytes and allogenic keratinocytes have been used,
i.e., keratinocytes autografting and keratinocyte
allografting. Although allografts do not survive
transplantation, they provide a biological dressing and
produce wound healing via various cytokine releases [35].
Melanocyte-containing epidermal grafts are an established
method for surgical management of leukodermas, e.g.,
piebaldism [36].
The development of keratinocyte culture has also led to an
increased understanding of factors influencing keratinocyte
growth and differentiation. Keratinocyte growth factor (KGF)
and GM-CSF released by dermal fibroblasts under the effect
of IL-1 α and β exert a paracrine effect on keratinocytes
and induce their proliferation and differentiation [37]. The
keratinocyte has been shown to play an active part in the
skin associated lymphoid tissue (SALT) both in cellular
interactions with Langerhans cells and epidermotropic T
lymphocytes and in the production of several growth factors
and cytokines (Table 1). Keratinocytes express Fc γ R and
this expression is upregulated by IFN γ [38]. Cytokines
produced by keratinocytes include interleukin (IL)-1, -6,
-7, -8, -10, -12, -15, -18, and -20 and TNF-α. Keratinocyte-derived
IL-7 and -15 are considered to be significant in T-cell
trafficking, possibly even in the pathogenesis of cutaneous
T-cell lymphoma. IL-15 inhibits keratinocyte apoptosis and
plays a role in psoriasis. Immunomodulatory IL-10 and -12
originating from keratinocytes are considered to be
responsible for systemic effects, and IL-18 perhaps has a
similar action. In addition, IL-12 suppresses ultraviolet
radiation-induced apoptosis by inducing DNA repair.
Keratinocytes were fairly recognized as being source or
target of other IL-10 family members like IL-20 and IL-24
and the role of these cytokines in specific diseases is
under investigation. In addition, a variety of cytokine
receptors are present on keratinocytes like those for IL-4,
-13, and -17 and to lesser degree IL-2. [39]. NO is produced
in high quantities by epidermal keratinocytes in psoriatic
skin in response to IFN- γ and TNF- α. Based on these
observations several biological agents are introduced in the
management of inflammatory skin conditions. Etanercept (Enbrel),
infliximab (Remicade) and adalimumab (Humira) as TNF-α
antagonists and anaknira (Kineret) as IL-1 blocker have been
recently approved for management of severe psoriasis,
psoriatic arthropathy and several other inflammatory
diseases [40].
Keratinocytes can be transformed by virus exposure, e.g.,
Simian virus 40 (SV40), human papilloma virus and by
oncogene transfections, which have illuminated the process
of skin carcinogenesis and antisense therapy to knock out
the mutant genes that are responsible for malignancy and
metastases. Keratinocytes have been transfected with human
growth hormone genes and have been shown to release human
growth hormone; a procedure that can be used in delivery of
gene products by genetically manipulated grafts [41]. In
this regard, this might form a basis for the use of
keratinocytes in gene therapy, perhaps to correct genetic
defects such as type VII collagen abnormalities in recessive
dystrophic epidermolysis bullosa. There is also a growing
interest in genetic manipulations ex vivo of epidermal
keratinocytes as a curative therapy for otherwise
untreatable hereditary dermatoses, such as mechanobullous
disorders and xeroderma pigmentosum [42]). Cultivated
keratinocytes have also been used to precisely localize and
define antigens targeted by various autoantibodies of
autoimmune bullous disorders by immunofluorescence,
immunoelectronmicroscopy and immunoblotting [43]. Nicotine
induced keratinization and cornification of cultured
keratinocytes, thus explaining leukokeratosis nicotinica
palati [44]). Human leukocyte elastase induces keratinocyte
proliferation in vitro, a finding that may explain
keratinocyte hyperproliferation in psoriasis [45]).
In keratinocytes, retinoids induce proliferation, resulting
in epidermal hyperplasia and modulate epidermal
differentiation [46]. Antineoplastic compounds exerted
growth inhibitory effect on malignant keratinocytes in
vitro. For example, perifosine, a novel alkylphospholipid,
induces p21 (WAF1) expression in squamous carcinoma cells
through a p53-independent pathway, leading to loss in cyclin-dependent
kinase activity and cell cycle arrest [47]). PUVA treatment
of keratinocytes reduced ICAM-1 and HLA-DR expression
induced by IFN γ and IL- α [48]. Vitamin D3 and its
analogues inhibited keratinocyte proliferation, which
explains the efficacy of these compounds in treatment of
hyperproliferative skin disorders, such as psoriasis [49].
It is now realized that the keratinocyte is not simply a
passive cell awaiting terminal differentiation but is an
active secretory cell with important biochemical and immune
functions.
Sebaceous Gland Cell (Sebocyte) Culture
Selective cultivation of normal human sebaceous gland
cells (sebocytes) has helped in investigating disorders of
the pilosebaceous units and served as a model for better
understanding of drug pharmacokinetics, such as retinoids,
androgens and toxic agents with special affinity to lipids
[50].
Sebocytes are specialized epithelial cells committed to
synthesize lipid rich sebum. Most of the available data of
the physiology and pathophysiology of the sebaceous gland
originates from hamsters [51]. However, several years ago in
vitro cultivation of pure adult human sebocytes has been
achieved and well characterized in MCDB 153 medium enriched
with serum, cholera toxin, crude BPE, bFGF, hydrocortisone
and insulin. Cells grew from the periphery of the lobules to
form a single layer sheet of polygonal cells resembling
basal keratinocytes (Fig. 5) that later undergo
differentiation with increase in cell volume, accumulation
of lipids and finally nuclear degeneration and cell death
(Fig. 6). Sebocytes in vitro show similar features to
keratinocytes being polygonal in shape and synthesize
cytokeratins, nevertheless they express specific epidermal
membrane antigen that has been used immunohistochemically in
their identification [52]. Analytical thin-layer
chromatography revealed that the cells synthesized a large
amount of sebum-specific lipids, squalene and wax esters in
vitro. As the lobules are obtained from adults, sebocyte
yield is usually low despite the several methods described
for sebocyte cultivation [53]. A modified technique
moderately improved the sebocyte yield, but obviously the
problem of the donor age and the necessity of serum
supplementation were unavoidable obstacles. To overcome the
yield problem, sebocytes were transfected by SV 40 large T
antigen and the immortalized sebaceous gland cell line SZ95
has been obtained that has been shown to preserve the major
functional and behavioral characteristics of normal human
sebocytes [54].
|
|
|
Fig. 5: Sebocytes in culture in the proliferative phase showing features of basal keratinocytes.
|
Fig. 6: Sebocytes in culture showing evidence of differentiation with refractile lipid vacuoles
|
It has also been shown that growth hormone stimulates
differentiation of sebocytes. Insulin-like growth hormone
(IGF-1) increases DNA synthesis and cell proliferation that
may contribute to the increase in sebum production during
puberty and in acromegaly [55]. Substance P, which can be
elicited by stress, promotes the development of cytoplasmic
organelles in sebaceous cells, stimulates sebaceous
germinative cells and increases the size of individual
sebaceous cells and the number of sebum vacuoles for each
differentiated sebaceous cell. Therefore, it seems that
substance P promotes both the proliferation and the
differentiation of sebaceous glands. It has been suggested
that this may be responsible for the stress-induced
exacerbation of acne [56]. The powerful sebosuppressive
effect of some retinoids, such as 13-cis-retinoic acid (isotretinoin),
demonstrates that sebocyte differentiation is altered by
retinoids. Retinoids inhibit proliferation and lipid
synthesis in cultured human sebocytes and alter their
keratin expression [57]. Later it has been shown that in
addition, 13-cis-retinoic acid (isotretinoin), 3,
4-didehydroretinoic acid, and 3, 4-didehydroretinol, but not
all-trans-retinoic acid (tretinoin) or the synthetic
retinoids acitretin and adapalene, were potent competitive
inhibitors of the oxidative 3 α –HSD activity of RoDH-4. As
a result, they reduce the formation of dihydrotestosterone
and androstandione in vitro that might explain the unique
sebosuppressive effect of isotretinoin when treating acne
[58]. Both testosterone and 5 α-dihydrotestosterone
significantly stimulated the proliferation of the sebocytes,
a finding that has been completely abolished by
spironolactone. Cultured sebocytes strongly expressed type 1
5 α-reductase and metabolized testosterone to
androstenedione, 5 α-androstanedione, 5 α-dihydrotestosterone,
androsterone and 5 α-androstanediol. Stimulation of sebocyte
proliferation by insulin, thyroid-stimulating hormone and
hydrocortisone indicates that the hormonal control of the
sebaceous gland could be a complex mechanism
[53, 59].
Incorporation of sebocytes and/or sebaceous glands in skin
reconstructs has not been achieved yet.
Langerhans Cell Culture Langerhans cells (LCs) are dendritic cells present in the
epidermis, bronchi and mucosae and are responsible for
antigen presentation. The essential role of LC in the
induction of contact allergic skin reactions and skin
transplant rejection is well established. They have also
been cultivated in vitro; however, they usually do not
survive more than few weeks as they are considered nearly
end differentiated cells and they are few in number.
Furthermore, properties of LC, such as phenotype, morphology
and the stimulatory potential to activate T lymphocytes, are
dependent on the local microenvironment in which the LCs
reside, thus correlation between in vitro and in vivo
findings is expectedly irrelevant. The initial use of
culture medium containing serum resulted in undue LCs
activation by serum antigens that further invalidated in
vitro findings [60].
Nevertheless, in vitro generation of dendritic cells (DCs)
with the typical molecular, morphological and functional
features of LC from purified CD34+ progenitor cells of
peripheral blood under defined serum-free culture conditions
has been achieved. TGF- β 1 was found to be an absolute
requirement for in vitro LC development under serum-free
conditions upon stimulation with the classical DC growth and
differentiation factors GM-CSF, TNF-α and SCF (Table 1). The
recently identified cytokine FLT3 ligand further
dramatically enhanced in vitro LC development and even
allowed efficient in vitro generation of LC colonies from
serum-free single cell cultures of CD34+ hemopoietic
progenitor cells (Fig. 7, 8) [61].
|
|
|
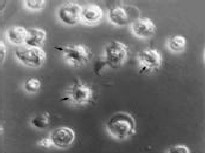
Fig. 7: Langerhans cells cultivated in the absence of serum. |
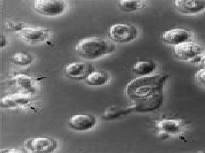
Fig. 8: Langerhans cells grown in presence of
serum showing higher number of dendritic cells. |
The procedure of LC cultivation was further refined by
isolation of immature epidermal LC in sufficient numbers
mechanically by three-step Ficoll floatation, a method that
was found to be superior to the well-known positive
immuno-magnetic separation by anti-CD1a-coated beads. This
has enabled easy isolation of LCs not only from peripheral
blood but also from the epidermis and mucosa. The medium
used is RPMI supplemented with HEPES, L-glutamine,
non-essential amino acids and sodium pyruvate. Both
epidermal and circulating LC appear rounded cells but with
multiple small dendrites [62].
Not only epidermal but also mucosal LCs have been cultivated
in vitro and it has been shown that LCs in human oral
epithelium are more efficient at stimulating T cells than
those of skin which may be of value as both cells meet
different antigenic challenges [63]. LCs could be
successfully kept viable after cryopreservation and it has
been shown that cryopreserved LCs expressed high levels of
HLA-DR and CD1a antigens and stimulated autologous T cells
to an extent almost identical to that obtained from fresh
LCs. These findings indicate that the cryopreservation of
human LCs could lead to a breakthrough in various
experiments dealing with these cells [64].
In atopy, Fc epsilon RI-activated LCs release chemotactic
signals and increase the migratory capacity of naive T cells
in vitro. Furthermore, they produce high amounts of
proinflammatory cytokines and chemokines, prime naive T
cells into IFN- γ -producing T cells and release IL-12 and
IL-18, which together might lead to the switch of the
initial T (H) 2-type immune response into a response of the
T (H) 1 type in vivo [65]. The role of LCs in contact
dermatitis has also been tested in vitro and it has been
found that contact sensitizers, such as dinitrochlorobenze,
caused an increase of the co-stimulatory molecule CD86, of
intercellular adhesion molecule CD54 and of the HLA-DR
antigen, whereas the irritant sodium dodecyl sulphate and
the vehicle dimethyl sulfoxide had no effect on LCs [66].
Recently, LCs have been used to test a variety of
immunomodulating agents in vitro. The topical
immunomodulators tacrolimus and pimecrolimus and steroid
ointment induced a selective depletion of inflammatory
dendritic cells in the epidermis of atopic dermatitis
patients and reduced the expression of the co-stimulatory
molecules CD80 and CD86 in vivo. However, tacrolimus did not
increase the rate of apoptotic LCs whereas steroid did so in
vitro [67]. Assessment of cellular parameters related to
UV-induced immunosuppression and the effect of sun screening
agents is performed using the reconstructed epidermis
containing LCs. Exposure to solar-simulated radiation
provokes morphological alterations, reduces surface antigen
expression and reduces the numbers of LCs cells within the
exposed epidermis [68].
At the present time there is a growing interest in the use
of cultivated LCs in the management of allergic contact
dermatitis and malignancy. Following activation by haptens,
LCs migrate to regional lymph nodes to activate effector T
cells. This step can be prevented in manipulated epidermal
LC by inhibitory peptide, 1 an apoptotic gene that binds and
blocks the function of hyaluronic acid (hyaluronan), which
is known to serve as an adhesive substrate for LC migration.
Furthermore, creating a "killer" LC clone by introduction of
CD95L cDNA, either before or after sensitization, results in
marked suppression of contact hypersensitivity by only
eliminating the pathogenic T cells [69]. The mitogenic
effect of TNF-α and GM-CSF on LCs in vitro has encouraged
physicians to use them systemically as an adjuvant therapy
to tumor vaccination [70]. Anti-melanoma immunization
therapy is currently tried in patients, through dendritic
cells charged with melanoma antigens with, so far, limited
side effects. Injections of antigenically charged dendritic
cells were performed subcutaneously, intravenously or in the
lymph nodes. Interestingly, positive clinical responses were
obtained with complete remission of the metastasis in some
cases [71].
Dermal Fibroblast Culture
Fibroblasts are the most numerous cells found in the loose
connective tissue and they are responsible for the
manufacture of all its elements or their precursors.
Fibroblasts are the easiest cells to cultivate in vitro, as
their growth requirements are minimal in comparison to other
human cell types. Indeed, their isolation and propagation
have helped not only in understanding the biology of these
cells but also the biology of other cells by serving as
control cells for almost all investigative work done so far.
They have also been used extensively in pharmacological
tests and skin reconstruction. Fibroblasts are grown in
DEMEM medium supplemented with L glutamine and 10% fetal
calf serum. They can also be adapted to grow in serum free
medium. When cultured in monolayer, dermal fibroblasts have
an elongated spindle shape with a prominent nucleus, are
small in diameter and grow at a high rate (Fig. 9)
[72].
Fibroblasts obtained from newborn generally have greater
mitogenic responsiveness than adult fibroblasts and that
age-associated loss of growth factor responsiveness may
contribute to the decreased proliferative capacity of
old-donor cells. It has been shown that dermal fibroblasts
possess a finite replicative capacity of 50 to 100
doublings, and then cease replication in response to growth
factors. Cells cultivated to the end of their replicative
life span in vitro overexpress metalloproteinase activities
that may explain the age-related progressive destruction of
the collagenous and elastic components of the extracellular
matrix [73]. In contrast to dermal fibroblasts, wound
fibroblasts grew slowly and were large, star shaped and had
cytoplasmic stress fibres. Smooth muscle α actin was
detected in the cytoplasm of most wound fibroblasts. Wound
fibroblasts also contract collagen gels during the first
days more strongly than dermal fibroblasts. These results
show that, in vitro, wound fibroblasts have greater
contractile capacity than dermal cells. The significant
proportion of wound fibroblasts containing α-smooth muscle
actin suggests that α-smooth muscle actin ratio may be
related to wound contraction [74].
|
|
|
Fig. 9: Fibroblasts in culture appear as spindle shaped
cells with indistinct cell boundaries. |
Fig.10: Endothelial cells in culture are similar
to fibroblasts but with distinct cellular boundaries |
Dermal fibroblasts are the source of collagen and elastin of
the extracellular matrix (ECM). Procollagens are secreted
through the Golgi apparatus in the extracellular space where
the N-terminal and C-terminal propeptides are cleaved by
specific proteases. The mature processed collagen molecules
aggregate to form larger collagen fibrils and help to form
the ECM with other components. Therefore, normal structural
and functional type I collagen production and deposition to
make normal physiological connective tissue needs regulation
at several steps. Abnormality in any step may cause hypo-,
hyper-, or defective synthesis and accumulation of collagen
in ECM, which in turn causes different diseases in humans,
such as osteogenesis imperfecta, scurvy, scleroderma or
systemic sclerosis, keloids, and others [75]. Elevated level
of type I collagen in scleroderma skin fibroblasts is
primarily due to the increased rate of collagen gene
transcription. Increasing evidence suggests that TGF- β
plays a significant role in fibrosis [76]. Dermal
fibroblasts are also an important source of several
cytokines, e.g., IL -1 and 6, chemokines, e.g.,
cyclooxygenase 2 (COX-2) and growth factors, e.g., FGF,
IGF-1 α that have significant autocrine and paracrine
effects (Table 1)[77]. Interferons α, β and γ suppress
collagen synthesis by dermal fibroblasts. In particular,
IFN- γ inhibits the constitutively increased collagen
synthesis characteristic of fibroblasts derived from lesions
of patients with scleroderma. Inhibition of collagen
synthesis by IFN- γ is associated with a coordinate
inhibition of transcription for types I and III collagen.
Animal studies demonstrated that IFN- γ inhibits the
collagen synthesis associated with the fibrotic response to
an implanted foreign body, bleomycin-induced pulmonary
fibrosis, and the healing response to cutaneous thermal
burns. Interferon α can decrease the size of keloids of
recent onset [78].
Hyaluronan is a ubiquitous extracellular matrix component,
and is present at high concentrations in skin, joints and
cornea. In the skin, it is synthesized primarily by dermal
fibroblasts and to a lesser extent by epidermal
keratinocytes. It is commonly used in cosmetics and skin
care preparations [79].
Dermal fibroblasts play also a significant role in
inflammation. They, besides neutrophils and macrophages, can
elaborate the proinflammatory matrix metalloproteinases 1, 3
and 9 in response to injurious agents, such as immune
complexes, lipopolysaccharides and UVB radiation both
directly and indirectly by IL-1 α and 6 released by
keratinocytes [80]. Dermal fibroblasts may contribute to the
epidermal hyperplasia of psoriasis by promoting keratinocyte
proliferation through IGF-1, the secretion of which could be
modulated by inflammatory cytokines, such as IFN-α (Table.
1) [81].
Several drugs have been tested with dermal fibroblasts. In
vitro exposure of fibroblasts to 8-methoxypsoralen and UVA
resulted in growth inhibition with morphological and
functional changes reminiscent of replicative senescence
that also occur in vivo upon exposure to sublethal
stressors, i.e., hyperoxia, hydrogen peroxide, and ethanol
[82]. Heparin inhibits dermal fibroblast proliferation and
collagen production but significantly stimulates bFGF and
TGF β 1 by normal, fetal and keloid fibroblasts which may
play a role in wound healing [83]. Although minocyclin has
been used to treat fibrosis, it did not alter dermal
fibroblast proliferation or collagen synthesis [84].
Retinoids stimulate dermal fibroblasts to produce
extracellular matrix proteins, particularly when the skin is
damaged by wounding, ultraviolet radiation or
glucocorticoids [85]. Inhibition of dermal activity by
cortisol in culture was partially reversed by testosterone
and dihydrotestosterone, whereas ACTH and the androgen
precursor dehydroepiandrosterone sulphate did not show such
antagonistic effect. These results suggest that increased
production of adrenal androgens during ACTH therapy may
account for the relative absence of 'skin-thinning' and
'steroid-bruising' which are common side-effects of
corticosteroid therapy [86]. On the other hand,
triamcinolone acetonide has been shown to decrease both
cellular proliferation and collagen production by dermal
fibroblasts and increases the production of bFGF and
decreases production of TGF- β 1 by human dermal fibroblasts
[87]. Glycolic acid (GA), commonly used in chemical peeling
and skin rejuvenation, increases collagen synthesis by
fibroblasts and modulates matrix degradation and collagen
synthesis through keratinocyte-released IL-1 α. These
results suggest that GA contributes to the recovery of
photodamaged skin through various actions, depending on the
skin cell type [88].
Besides the use of dermal fibroblasts in almost all skin
substitutes, there are currently trials attempting at
transplanting genetically modified fibroblasts carrying a
stably integrated tarnsgenes as a vehicle for normal genes
[89].
Dermal Endothelial Cell Culture
Dermal endothelial cells involved in tumor angiogenesis,
wound healing, and inflammation are predominantly of micro
vascular origin and are functionally distinct from large
vessel-derived endothelial cells which have been largely
used for in vitro vascular research. Initially, cultivation
of dermal endothelial cells has been achieved using high
concentrations of serum and conditioned media from tumors to
achieve optimal growth [90]. Contamination of the cultures
with other cell types, such as fibroblasts was evident,
however, with some modifications of the culture procedure in
which continuous Percoll gradient was applied; pure
endothelial cells could be obtained. The culture medium
consisted of endothelial cell basal medium supplemented with
EGF and hydrocortisone [91]. The cultivation of endothelial
cells in vitro was later facilitated by the incorporation of
the potent mitogen vascular endothelial growth factor (VEGF)
that also functions as a survival factor for endothelial
cells by up-regulating Bcl-2 expression (Fig. 10) [92].
Several cellular components of the skin synthesize and
release VEGF in vivo and in vitro (Table 1).
Clearly, the growth and turnover of endothelial cells in the
skin is fundamental not only in normal development, but also
in wound repair, hair follicle cycling, tumor cell
metastasis, and in many different states of cutaneous
pathology. Endothelial cells in vitro acquire a slightly
elongated epithelioid (cobblestone) shape in the modified
and now commercially available endothelial cell growth
medium MV and are labeled with Ulex europaeus Agglutinin I
and an antibody against Factor VIII-related antigen [93]. To
further overcome the problems commonly involved in the
culture of microvascular endothelial cells, including
unreliable isolation techniques and low cell yields, a
simplified protocol for the selective isolation and
cultivation of human dermal microvascular endothelial cells
from neonatal foreskins has been proposed. It utilizes
immunomagnetic beads and it is based on the transient,
endothelial cell-specific induction of E-selectin by TNF-α
[94].
The effects of several cytokines have been examined on
cultivated dermal endothelial cells in vitro. GM-CSF and
IL–1 α and β induced endothelial cell proliferation, whereas
IFN– γ and TNF α activated them by induction of HLA-DR and
ICAM-1 expression [91]. Activated endothelial cells also
help in initiating immune reactions as antigen presenting
cells as they express adhesion molecules such as E selectin,
ICAM- 1 and VCAM-1 that are essential in memory and effector
T cell recruitment [95]. In wound healing, a dynamic
interaction occurs among endothelial cells, angiogenic
cytokines, such as FGF, VEGF, TGF- β, angiopoietin, and mast
cell tryptase, and the extracellular matrix environment
[96]. Fibrin fragment E liberated at wounds stimulates the
proliferation, migration and differentiation of human dermal
microvascular endothelial cells in vitro, both in the
absence and presence of additional endothelial growth
factors, such as VEGF and bFGF indicating a possible synergy
between the signaling pathways used by these three
angiogenic factors [97]. Increase of Bcl 2 on endothelial
cells is associated with intratumoral angiogenesis
enhancement and accelerated tumor growth [98]. On the other
hand, several agents inhibited endothelial cell
proliferation and angiogenesis. Angiotensin type II (AT2)
receptor stimulation is antiangiogenic through inhibition of
VEGF-induced endothelial cell migration and tube formation
via activation of a PTX-sensitive G protein [99].
Kaposi's sarcoma-associated herpes virus (KSHV) is
consistently found in all forms of Kaposi’s sarcoma.
Infection of dermal microvascular endothelial cells with
KSHV induces spindle cell formation in vitro by a c-kit
dependent mechanism. Endothelial cells are used as host
cells for culturing and propagation of this virus [100].
Similarly, Bartonellosis, a disease caused by motile
intracellular bacteria, produces characteristic dermal
eruption that results from a pronounced endothelial cell
proliferation (Verruga peruana). Also an extract of
Bartonella bacilliformis organisms induced endothelial cell
proliferation in vitro [101].
The effect of physical agents and drugs has also been
studied on endothelial cells in vitro. Hypoxia resulted in
synthesis and release of VEGF by endothelial cells and
possibly by pericytes which contributes to their
proliferation and subsequent angiogenesis, i.e., autocrine
effect [102]. Ultraviolet radiation induces nitric oxide
synthase 2 expression by endothelial cells which may provide
an explanation of the erythema, edema and inflammation
[103]. On the other hand, hypertension in end stage renal
failure has been attributed to uremic plasma factors
inhibiting nitric oxide synthase 2 with subsequent lack of
vasodilatation and persistent vasoconstriction [104]. Tumors
promote angiogenesis by secreting growth factors, such as
VEGF and VEGF-related molecules either spontaneously or in
response to hypoxia that stimulates endothelial migration,
proliferation, proteolytic activity, and capillary
morphogenesis. Newly formed blood vessels supply the tumor
with nutrients and oxygen, dispose of its metabolic waste
products, and generate paracrine stimuli, which further
promote tumor cell proliferation and invasiveness [105].
Endostatin exerts it powerful antiangiogenic effect through
inhibition of endothelial cell proliferation by E selectin
dependent mechanism [106].
Griseofulvin has been used as an antifungal drug for many
years, but it has recently been shown to be effective in
several inflammatory skin diseases. Griseofulvin inhibited
the expression of TNF α-induced VCAM-1 dose-dependently, and
this inhibition was fully reversible. Similarly,
griseofulvin inhibited the induction of VCAM-1 expression on
both TNF α- and IL-1 α-stimulated human endothelial cells
indicating potent immunomodulatory properties, which may be
associated with its feature as a microtubule antagonist
[107]. Retinoids inhibit whereas steroids induce
proliferation of endothelial cells without effecting HLA-DR,
ICAM-1 induction by IFN γ, TNF α and IL-1 α [108].
Nevertheless, retinoids inhibited VCAM-1 gene expression and
VCAM-1 T cell binding to cytokine-treated endothelial cells
thus explaining their anti-inflammatory effect [109].
Successful reconstruction of human skin model containing
keratinocytes, fibroblasts and endothelial cells has been
described. The therapeutic implications of this culture have
yet not been verified [101].
Hair Follicle Cell Cultures
Different culture models are used to investigate hair
biology. With organ cultures the whole hair follicle is
maintained in the culture in order to investigate the effect
of different substances on hair growth. The development of
medications to stimulate or to inhibit the production of
hair keratine may have great impact on the treatment of
baldness and hypertrichosis, respectively [111].
Regarding selective cell culture models, two different
compartments of the hair follicle are the main objective of
research, namely hair keratinocytes from the outer root
sheath (ORS) and mesenchymal cells of dermal papilla (DPC).
ORS keratinocytes can be easily obtained from plucked hairs,
using the trichogramm technique [112], thus avoiding any
surgical procedure to isolate the ORS keratinocytes. Culture
medium and mitogens are almost the same that are used in
selective cultivation of epidermal keratnocytes. With this
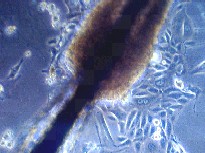
culture model the outgrowth of keratinocytes from the ORS is
seen as early as two days following inoculation (Fig. 11);
they produce multilayered cell colonies, in which desmosomes
could be identified [112]. Alltough they do not produce the
hair keratin, these cells can be used to study hair biology
and indeed the inhibition of androgens on ORS keratinocytes
proliferation has already been shown [113]. Vellus hair
follicle cells were also successfully cultivated [114].
|

|
|
Figure 11: Outgrowth of keratinocytes
from the outer root sheath. |
Figure 12:Typical aggregative growth
pattern of dermal papilla cells |
As a source of keratinocytes and similar to epidermal
cultures, ORS cells can also be used to treat recurrent leg
ulcers [115]. Future models in this field may elucidate
important aspects of hair graying and even may have
therapeutic implications, since melanocytes of graying and
white hair follicles can be induced to produce melanin in
vitro [116].
DPC have a close contact to the keratin-producing hair
matrix cells, being very important to study
epithelial-mesenchymal interactions in the hair follicle.
Complex culture-models have been developed [117]. They can
be dissected from the hair follicle and grow in medium
identical to that of dermal fibroblasts [118]. They have
gained much interest due to their ability to induce hair
growth after reimplantation in the skin
[119,120].
When DPC were compared to dermal fibroblasts, many
differences were found. They express muscle antigens [121],
have a weaker capacity to contract collagen lattices and
have an aggregative growth pattern [122] (Figure 12).
Recently a difference in expression and activation of matrix
metalloproteinases was also shown [123], reinforcing the
concept that they are a specialized subpopulation of
mesenchymal cells.
As a target of androgen effect in the hair follicle
[113],124]
, DPC were used to investigate androgenetic alopecia
[125]. Well stablished treatments for this condition were
confirmed in vitro using DPC. Minoxidil has a proliferative
and anti-apoptotic effect [126] and finasteride increases
the expression of growth factors on DPC in vitro [127]
Other Cutaneous Cell Cultures
Other cutaneous cells of less importance have also been
cultivated in vitro. Merkel cell is a highly specialized
cell of neural crest origin that primarily acts as slowly
adapting mechanoreceptors. Their in vitro cultivation and
characterization has been hampered by their paucity, lack of
information about specific mitogens and their state of
differentiation. Nevertheless, they have recently been
cultivated from animals and humans from both glabrous and
hairy skin [128]. Merkel cells of sinus hair follicles of
rats have been grown as a monolayer that exhibited flat
round morphology with extended lamellae on their fringes in
serum free medium, whereas they acquire a fibroblast-like
morphology and survive longer in the presence of serum
[129]. Establishment of a cell line from Merkel cell
carcinoma in humans was possible. In general, cells grow as
loosely arranged floating small aggregates on RPMI medium on
irradiated fibroblast feeder layer. They also retain their
in vivo properties, namely expression of cytokeratin 18,
neuron-specific enolase, neurofilaments and synaptophysin.
The expression of the distinct cytokeratin 20 has greatly
facilitated their identification in vitro and in vivo and
also improved detection of micro metastasis of Merkel cell
carcinoma [130].
Epithelial cells were also cultivated from the secretory
coil and collecting ducts of the sweat glands in a medium
supplemented with fetal calf serum, insulin, transferring,
epidermal growth factor and hydrocortisone. Initially, cells
of the secretory coil were elongated while of the collecting
ducts were polyhedral [131]. Fibroblast overgrowth was
markedly diminished by dispase separation and use of serum
free medium similar to keratinocytes. Cells expressed
carcinoembryonic antigen, K 8.37 and K 8.13 [132].
Ultrastructural studies revealed domes indicative of
transepithelial active ion transport in cells of
reabsorptive duct [131]. Sweat gland myoepithelial cells in
culture expressed, in addition to unique cytokeratins, α
smooth muscle actin (sma) [133]. Cells isolated from
patients with cystic fibrosis were morphologically
indistinguishable from normal except for high
transepithelial resistance and increased amiloride
sensitivity, i.e., functionally abnormal [134]. Skin
equivalent studies revealed that keratinocytes of young
donor could differentiate toward sweat ducts in vitro in the
presence of serum, EGF and dermal fibroblasts [135]135).
References
Introduction
1. Cruickshank CN, Cooper JR, Hooper C. The cultivation of cells from adult epidermis. J Invest Dermatol 1960, 34:339-342.
2. Jansen B, Zangemeister-Wittke U. Antisense therapy for cancer-the time of truth. Lancet Oncol 2002; 3:672-683.
3. Fusenig NE. Epithelial-mesenchymal interactions regulate keratinocyte growth and differentiation in vitro. In: The Keratinocyte Handbook (Leigh, I. et al., eds). Cambridge University Press. 1994, pp 71-94.
4. Stark H-J. Bauer M, Breitkreutz D, Mirancea N, Fusenig NE. Organotypic keratinocyte coculture in defined medium with regular epidermal morphogenesis and differentiation. J Invest Dermatol 1999, 112, 681-691.
5. Stark H-J. Szabowski A, Fusenig NE, Maas-Szabowski N.Organotypic co-cultures as skin equivalents: A complex and sophisticated in vitro system. Biol Proced Online 2004, 6:55-60.
Melanocyte Culture
6. Gilchrest BA, Vrabel MA, Flynn E, Szabo G. Selective cultivation of human melanocytes from newborn and adult epidermis. J Invest Dermatol 1984; 83:370-376.
7. Eisinger M, Marko O. Selective proliferation of normal human melanocytes in vitro in the presence of phorbol ester and cholera toxin. Proc Natl Acad Sci USA 1982, 79:2018-2022.
8. Seiberg M. Keratinocyte-melanocyte interactions during melanosome transfer. Pigment Cell Res 2001; 14:236-242.
9. Abdel-Naser MB: Mitogen requirements of normal human melanocytes in a serum and tumor promoter free medium. Eur J Dematol 2003: 13:29-33.
10. Abdel-Naser MB: Differential effects on melanocyte growth and melanization of low vs high Ca++ keratinocyteconditioned medium. Brit J Dermatol 1999; 140:50-55.
11. Gilchrest BA, Treloar V, Grassi AM, Yaar M, Szabo G, Flynn E. Characteristics of adult human nevocellular nevus cells. J Invest Dermatol 1986; 87:102-106.
12. Tada A, Periera E, Beitner-Jihnson D, Kavaungh R, Abdel-Malek ZA. Mitogen and ultraviolet B-induced signaling pathways in normal human melanocytes. J Invest Dermatol 2002; 118:316-322.
13. Abdel-Naser MB, Krasagakis K, Garbe C, Eberle J. Direct effects on proliferation, antigen expression and melanin synthesis of cultured normal human melanocytes in response to UVB and UVA light. Photodermatol Photoimmunol Photomed 2003; 19:122-127.
14. Cross KJ, Mustoe TA. Growth factors in wound healing. Surg Clin North Am 2003; 83:531-545.
15. Halaban R. The regulation of normal melanocyte proliferation. Pigment Cell Res 2000; 13:4-14.
16. Imokawa G. Autocrine and paracrine regulation of melanocytes in human skin and in pigmentary disorders. Pigment Cell Res 2004; 17:96-110.
17. Van den Wijngaard R, Wankowicz-Kalinska A, Pals S, Weening J, Das P. Autoimmune melanocyte destruction. Lab Invest 2001; 81:1061-1067.
18. Abdel-Naser MB, Blume U, Abdel-Fattah A, Orfanos CE.Evidence for a complement mediated inhibition and an antibody dependent cellular cytotoxicity of dermal fibroblasts in alopecia areata. Acta Derm Vener (Stockh)1994; 74:351-354.
19. Iuga AO, Qureshi AA, Lerner EA. Nitric oxide is toxic to melanocytes in vitro. Pigment Cell Res. 2004; 17:302-306.
20. Kasraee B, Handjani F, Aslani FS. Enhancement of the depigmenting effect of hydroquinone and 4-hydroxyanisole by all-trans-retinoic acid (tretinoin): the impairment of glutathione-dependent cytoprotection? Dermatology 2003; 206:289-291.
21. Yoon TJ, Lei TC, Yamaguchi Y, Batzer J, Wolber R, Hearing VJ. Reconstituted 3-dimensional human skin of various ethnic origins as an in vitro model for studies of pigmentation. Anal Biochem 2003; 318:260-269.
22. Abdel-Naser MB, Hann SK, Bystryn JC. Oral psoralen with UVA therapy releases circulating growth factor(s) that stimulate cell proliferation. Arch Dermatol 1997; 133:1530-1533.
23. Duval C, Schmidt R, Regnier M, Facy V, Asselineau D, Bernerd F. The use of reconstructed human skin to evaluate UV-induced modifications and sunscreen efficacy. Exp Dermatol 2003; 12:64-70.
24. Perlis C, Herlyn M. Recent advances in melanoma biology.The Oncologist 2004; 9:182-187.
25. Guerra L, Primavera G, Raskovic D, Pellegrini G, Golisano O, Bondanza S, Paterna P, Sonego G, Gobello T, Atzori F, Piazza P, Luci A, De Luca M. Erbium:YAG laser and cultured epidermis in the surgical therapy of stable vitiligo. Arch Dermatol 2003; 139:1303-1310.
Keratinocyte Culture
26. Rheinwald JG, Green H. Epidermal growth factor and multiplication of cultured human keratinocytes. Nature 1977; 265:421-424.
27. Holbrook KA, Hemmings H. Phenotypic expression of epidermal cell in vitro; a review. J Invest Dermatol 1983; 81:11-24.
28. Boyce ST, Ham RG. Calcium regulated differentiation of normal human epidermal keratinocytes in chemically defined clonal culture and serum-free serial culture. J Invest Dermatol 1983; 81:33-40.
29. Albers KM, Taichmann LB. Kinetics of withdrawal from the cell cycle in cultured human epidermal keratinocytes. J Invest Dermatol 1984; 82:161-164.
30. Abdel-Naser MB, Abdel-Fattah A, Abdallah MA. Selective cultivation of epidermal keratinocytes from newborn and adult skin in a serum free medium and without the use of a feeder layer. Gulf J of Dermatol and Venereol 1998; 5:18-23.
31. Wollina U, Heide M, Mueller-Litz W, Obenauf D, Ash J. Functional textiles in prevention of chronic wounds, wound healing and tissue engineering. Current Problems of Dermatology 2003; 31:82-96.
32. Gallico GG, O’Connor NE, Compton CC, Remensnyder JP, Kehinde O, Green H. cultured epithelial autografts for giant congenital nevi. Plast Reconstr Surg 1989; 84:1-9.
33. De Luca M, Albanese E, Cancedda R, Viacava A, Faggiuni A, Zambruno G, Giannetti A. Treatment of leg ulcers with cryopreserved allogenic cultured epithelium. Arch Dermatol 1992; 128:633-638.
34. Romagnoli G, De Luca M, Faranada F, Bandelloni R, Franzi AT, Cataliotti F, Cancedda R. treatment of posterior hypospadius by autologous graft of cultured urethral epithelium. N Eng J Med 1990; 323:527-530.
35. Ehrlich HP Understanding experimental biology of skin equivalent: from laboratory to clinical use in patients with burns and chronic wounds. Am J Surg 2004; 187:29-33.
36. Guerra L, Primavera G, Raskovic D, Pellegrini G, Golisano O, Bondanza S, Kuhn S, Piazza P, Luci A, Atzori F, De Luca M. Permanent repigmentation of piebaldism by erbium:YAG laser and autologous cultured epidermis. Br J Dermatol 2004; 150:715-721.
37. Werner S, Smola H. Paracrine regulation of keratinocyte proliferation and differentation. Cell Biol 2001; 11:143-146.
38. Cauza K, Grassauer A, Hinterhuber G, Horvat R, Rappersberger K, Wolff K, Foedinger D. FcgammaRIII expression on cultured human keratinocytes and upregulation by interferon-gamma. J Invest Dermatol 2002; 119:1074-1079.
39. Grone A. Keratinocytes and cytokines.Vet Immunol Immunopathol. 2002; 88:1-12.
40. http://www.medscape.com/view article/463653. Shaun Ruddy. Cytokine blockers for inflammatory arthritis.
41. Morgan JR, Barrandon Y, Green H, Mulligan RC. Expression of an exogenous growth hormone gene by transplantable human epidermal cells. Science 1987; 237: 1476-1479.
42. Magnaldo T, Sarasin A. Genetic reversion of inherited skin disorders. Mutat Res. 2002; 509:211-220.
43. Batteux F, Franck N, Jaffray P, Raulo M, Prost C, Leibowitch M, Escande JP, Weill B. An extract from cultured human keratinocytes that contains the major autoantigens related to autoimmune bullous skin diseases. J Clin Immunol 1997; 17:228-233.
44. Groemer FE, Bernd A, Bereiter-Hahn J, Holzmann H. A new model of epidermal differentation: induction by mechanical stimulation. Arch Dermatol Res 1990; 282:22-32.
45. Rogalski C, Meyer-Hoffert U, Proksch E, Wiedow O: Human leukocyte elastase induces keratinocyte proliferation in vitro and in vivo. J Invest Dermatol 2002; 118:49-54.
46. Gendimenico GJ, Mezick JA. Pharmacological effects of retinoids on skin cells. Skin Pharmacol 1993; 6:24-34.
47. Patel V, Lahusen T, Sy T, Sausville EA, Gutkind JS, Senderowicz AM. Perifosine, a novel alkylphospholipid, induces p21 (WAF1) expression in squamous carcinoma cells through a p53-independent pathway, leading to loss in cyclin-dependent kinase activity and cell cycle arrest. Cancer Res 2002; 62:1401-1409.
48. Urano R, Urano K, Matsuyama T, Matsuo I, Ohkido M PUVA treatment of human keratinocytes suppresses the surface expression of intercellular adhesion molecule 1 (ICAM-1) and inhibits adherence of PHA-blasts in vitro. Tokai J Exp Clin Med 1997; 22:71-76.
49. Kobayashi T, Hashimoto K, Yoshikawa K. Growth inhibition of human keratinocytes by a new vitamin D3 analogue in vitro. J Dermatol Sci 1998; 16:158-164.
Sebaceous Gland Cell (Sebocyte) Culture
50. Cunliffe WJ. The sebaceous gland and acne- 40 years on. Dermatology 1998; 196:9-15.
51. Ito A, Sakiguchi T, Kitamura K, Akamatsu H, Horio T. Establishment of a tissue culture system for hamster sebaceous gland cells. Dermatology 1998; 197:238-244.
52. Xia L, Zouboulis Ch, Detmar M, Mayer-de-Silva A, Stadler R, Orfanos CE. Isolation of human sebaceous glands and cultivation of sebaceous gland derived cells as an in vitro model. J Invest Dermatol 1989; 93:315-321.
">53. Fujie T, Shikiji T, Uchida N, Urano Y, Nagae H, Arase S.Culture of cells derived from the human sebaceous gland under serum-free conditions without a biological feeder layer or specific matrices. Arch Dermatol Res 1996; 288:703-708.
54. Zouboulis ChC, Seltmann H, Neitzel H, Orfanos CE. Establishment and characterization of an immortalized human sebaceous gland cell line (SZ95). J Invest Dermatol 1999; 113:1011-1020.
55. Deplewski D. Rosenfield RL. Growth hormone and insulin-like growth factors have different effects on sebaceous cell growth and differentiation. Endocrinology 1999; 140:4089– 4094.
56. Toyoda M, Morohashi M. Pathogenesis of acne. Med Electron Microsc 2001; 34:29-40.
57. Gendimenico GJ, Mezick JA. Pharmacological effects of retinoids on skin cells. Skin Pharmacol 1993; 6:24-34.
58. Karlsson T, Vahlquist A, Kedishvili N, Torma H. 13-cis-retinoic acid competitively inhibits 3 alpha-hydroxysteroid oxidation by retinol dehydrogenase RoDH-4: a mechanism for its anti-androgenic effects in sebaceous glands? Biochem Biophys Res Commun 2003; 303:273-278.
59. Zouboulis CC, Xia L, Akamatsu H, Seltmann H, Fritsch M, Hornemann S, Ruhl R, Chen W, Nau H, Orfanos CE. The human sebocyte culture model provides new insights into development and management of seborrhea and acne. Dermatology 1998; 196:21-31.
Langerhans Cell Culture
60. Teunissen MB. Dynamic nature and function of epidermal Langerhans cells in vivo and in vitro: a review, with emphasis on human Langerhans cells. Histochem J 1992; 24:697-716.
61. Strobl H, Riedl E, Bello-Fernandez C, Knapp W. Epidermal Langerhans cell development and differentiation. Immunobiology. 1998; 198: 588-605.
62. Tchou I, Sabido O, Lambert C, Misery L, Garraud O, Genin C. Technique for obtaining highly enriched, quiescent immature Langerhans cells suitable for ex vivo assays. Immunol Lett 2003; 86: 7-14 .
63. Hasseus B, Jontell M, Bergenholtz G, Dahlgren UI. Langerhans cells from human oral epithelium are more effective at stimulating allogeneic T cells in vitro than Langerhans cells from skin. Clin Exp Immunol. 2004; 136:483-489.
64. Seo KI, Huh CH, Han JH, Youn JI, Lee CH, Lee WJ, Eun HC. Characterization of cryopreserved human Langerhans cells. Cryobiology. 2002; 45: 118-126.
65. Novak N, Valenta R, Bohle B, Laffer S, Haberstok J, Kraft S, Bieber T. FcepsilonRI engagement of Langerhans cell-like dendritic cells and inflammatory dendritic epidermal cell-like dendritic cells induces chemotactic signals and different T-cell phenotypes in vitro. J Allergy Clin Immunol. 2004; 113: 949-957
66. Tuschl H, Kovac R. Langerhans cells and immature dendritic cells as model systems for screening of skin sensitizers. Toxicol In Vitro 2001; 15: 327-331.
67. Schuller E, Oppel T, Bornhovd E, Wetzel S, Wollenberg A. Tacrolimus ointment causes inflammatory dendritic epidermal cell depletion but no Langerhans cell apoptosis in patients with atopic dermatitis. J Allergy Clin Immunol 2004; 114:137-143 .
68. Duval C, Schmidt R, Regnier M, Facy V, Asselineau D, Bernerd F. The use of reconstructed human skin to evaluate UV-induced modifications and sunscreen efficacy. Exp Dermatol 2003; 12: 64-70.
69. Takashima A, Mummert M, Kitajima T, Matsue H. New technologies to prevent and treat contact hypersensitivity responses. Ann N Y Acad Sci 2000; 919: 205-213 .
70. Janik JE, Miller LL, Kopp WC, Taub DD, Dawson H, Stevens D, Kostboth P, Curti BD, Conlon KC, Dunn BK, Donegan SE, Ullrich R, Alvord WG, Gause BL, Longo DL. Treatment with tumor necrosis factor-alpha and granulocyte-macrophage colony-stimulating factor increases epidermal Langerhans' cell numbers in cancer patients. Clin Immunol 1999; 93:209-221.
71. Schmitt D. The Langerhans cell: from in vitro production to use in cellular immunotherapy. J Soc Biol. 2001; 195:69-74.
Dermal Fibroblast Culture
72. Layman DL, Mc Goodwin EB, Martin GR. The nature of the collagen synthesized by cultured human fibroblasts. Proc Natl Acad Sci USA 1971; 74: 454-458
73. West MD. The cellular and molecular biology of skin aging. Arch Dermatol 1994; 130: 87-95
74. Moulin V, Castilloux G, Jean A, Garrel DR, Auger FA, Germain L. In vitro models to study wound healing fibroblasts. Burns 1996; 22: 359-362
75. Uitto J, Kouba D. Cytokine modulation of extracellular matrix gene expression: relevance to fibrotic skin diseases. J Dermatol Sci 2000; 24:S60–S69
76. Ghosh AK. Factors Involved in the Regulation of Type I Collagen Gene Expression: Implication in Fibrosis. Exp Biol Med 2002; 227:301–314
77. Kessler-Becker D, Krieg T, Eckes B. Expression of pro-inflammatory markers by human dermal fibroblasts in a three-dimensional culture model is mediated by an autocrine interleukin-1 loop. Biochem J 2004; 379: 351-358.
78. Granstein RD, Flotte TJ, Amento EP. Interferons and collagen production. J Invest Dermatol 1990; 95:75-80
79. Kakehi K, Kinoshita M, Yasueda S. Hyaluronic acid: separation and biological implications. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 797:347-355
80. Warner RL, Bhagavathula N, Nerusu KC, Lateef H, Younkin E, Johnson KJ, Varani J. Matrix metalloproteinases in acute inflammation: induction of MMP-3 and MMP-9 in fibroblasts and epithelial cells following exposure to pro-inflammatory mediators in vitro. Exp Mol Pathol 2004; 76:89-95
81. Miura H, Sano S, Higashiyama M, Yoshikawa K, Itami S.Involvement of insulin-like growth factor-I in psoriasis asa paracrine growth factor: dermal fibroblasts play a regulatory role in developing psoriatic lesions. Arch Dermatol Res 2000; 292:590-597
82. Wlaschek M, Ma W, Jansen-Durr P, Scharffetter-Kochanek K. Photoaging as a consequence of natural and therapeutic ultraviolet irradiation-studies on PUVA-induced senescence-like growth arrest of human dermal fibroblasts.Exp Gerontol. 2003; 38:1265-1270
83. Carroll LA, Koch RJ. Heparin stimulates production of bFGF and TGF-beta 1 by human normal, keloid, and fetal dermal fibroblasts. Med Sci Monit 2003; 9: 97-108
84. Anderegg U, Prieb J, Hildebrandt G, Saalbach A. Minocycline does not alter collagen type I metabolism of dermal fibroblasts in culture. Arch Dermatol Res 2002; 294:103-108
85. Gendimenico GJ, Mezick JA. Pharmacological effects of retinoids on skin cells. Skin Pharmacol 1993; 6: 24-34
86. Harvey W, Grahame R, Panayi GS. Effects of steriod hormones on human fibroblasts in vitro. II. Antagonism by androgens of cortisol-induced inhibition.Ann Rheum Dis 1976; 35:148-151
87. Carroll LA, Hanasono MM, Mikulec AA, Kita M, Koch RJ. Triamcinolone stimulates bFGF production and inhibits TGF-beta1 production by human dermal fibroblasts. Dermatol Surg 2002; 28: 704-709.
88. Okano Y, Abe Y, Masaki H, Santhanam U, Ichihashi M, Funasaka Y. Biological effects of glycolic acid on dermal matrix metabolism mediated by dermal fibroblasts and epidermal keratinocytes. Exp Dermatol 2003; 12: 57-63.
89. Krueger GG. Fibroblasts and dermal gene therapy: a minireview. Hum Gene Ther. 2000; 11: 2289-2296.
Dermal Endothelial Cell Culture
90. Davison P, Bensch K, Karasek M. Isolation and long term serial cultivation of endothelial cells from the microvessels of the adult human dermis. In Vitro 1983; 19:937-945
91. Ruszczak Zb, Detmar M, Imke E, Orfanos. Effects of rIFN-alpha,-beta and –gamma on the morphology, proliferation and cell surface antigen expression of human dermal microvascular endothelial cells in vitro. J Invest Dermatol 1990; 95: 693-699
92. Ferrara N, Davis-Smyth T: The biology of vascular endothelial growth factor. Endocr Rev 1997; 18: 4–25
93. Imcke E, Ruszczak Z, Mayer-da Silva A, Detmar M, Orfanos CE. Cultivation of human dermal microvascular endothelial cells in vitro: immunocytochemical and ultrastructural characterization and effect of treatment with three synthetic retinoids. Arch Dermatol Res 1991; 283: 149-157
94. Richard L, Velasco P, Detmar M. A simple immunomagnetic protocol for the selective isolation and long-term culture of human dermal microvascular endothelial cells. Exp Cell Res 1998; 240:1-6
95. Pober JS, Kluger MS, Schechner JS. Human endothelial cell presentation of antigen and the homing of memory/effector T cells to skin. Ann N Y Acad Sci 2001; 941:12-25
96. Tonnesen MG, Feng X, Clark RA. Angiogenesis in wound healing. J Investig Dermatol Symp Proc 2000; 5: 40-46
97. Bootle-Wilbraham CA, Tazzyman S, Thompson WD, Stirk CM, Lewis CE. Fibrin fragment E stimulates the proliferation, migration and differentiation of human microvascular endothelial cells in vitro. Angiogenesis 2001; 4:269-275
98. Nor JE, Christensen J, Liu J, Peters M, Mooney DJ, Strieter RM, Polverini PJ. Up-Regulation of Bcl-2 in microvascular endothelial cells enhances intratumoral angiogenesis and accelerates tumor growth. Cancer Res 2001; 61: 2183-2188
99. Benndorf R, Boger RH, Ergun S, Steenpass A, Wieland T. Angiotensin II type 2 receptor inhibits vascular endothelial growth factor-induced migration and in vitro tube formation of human endothelial cells. Circ Res 2003; 93:438-447
100. Moses AV, Jarvis MA, Raggo C, Bell YC, Ruhl R, Luukkonen BG, Griffith DJ, Wait CL, Druker BJ, Heinrich MC, Nelson JA, Fruh K. A functional genomics approach to Kaposi's sarcoma. Ann NY Acad Sci 2002; 975:180-191
101. Garcia FU, Wojta J, Broadley KN, Davidson JM, Hoover RL: Bartonella bacilliformis stimulates endothelial cells in vitro and is angiogenic in vivo. Am J Pathol 1990; 36:1125-1135
102. Yonekur H, Sakurai S, Liu X, Migita H, Wang H, Yamagishi S-I, Nomura M. Abedin J, Unoki H, Yamamoto Y, Yamamoto H. Placenta growth factor and vascular endothelial growth factor B and C expression in microvascular endothelial cells and pericytes. J Biol Chem 1999; 274:35172-35178
103. Suschek CV, Bruch-Gerharz D, Kleinert H, Forstermann U, Kolb-Bachofen V. Ultraviolet A1 radiation induces nitric oxide synthase-2 expression in human skin endothelial cells in the absence of proinflammatory cytokines. J Invest Dermatol 2001;117: 1200-1205
104. Xiao S, Schmidt RJ, Baylis C. Plasma from ESRD patients inhibits nitric oxide synthase activity in cultured human and bovine endothelial cells. Acta Physiol Scand 2000; 168:175-179
105. Nicosia RF. What is the role of vascular endothelial growth factor-related molecules in tumor angiogenesis? Am J Pathol 1998; 153: 11-16
106. Yu Y, Moulton KS, Khan MK, Vineberg S, Boye E, Davis VM, O'Donnell PE, Bischoff J, Milstone DS. E-selectin is required for the antiangiogenic activity of endostatin. Proc Natl Acad Sci USA 2004; 101: 8005-8010
107. Asahina A, Tada Y, Nakamura K, Tamaki K. Griseofulvin has a potential to modulate the expression of cell adhesion molecules on leukocytes and vascular endothelial cells. Int Immunopharmacol 2001; 1: 75-83
108. Hettmannsperger U, Tenorio S, Orfanos CE, Detmar M. Corticosteroids induce proliferation but do not influence TNF- or IL-1 beta-induced ICAM-1 expression of human dermal microvascular endothelial cells in vitro. Arch Dermatol Res 1993; 285: 347-351
109. Gille J, Paxton LL, Lawley TJ, Caughman SW, Swerlick RA. Retinoic acid inhibits the regulated expression of vascular cell adhesion molecule-1 by cultured dermal microvascular endothelial cells. J Clin Invest 1997; 99:492-500
110. Sahota PS, Burn JL, Heaton M, Freedlander E, Suvarna SK, Brown NJ, Mac Neil S. Development of a reconstructed human skin model for angiogenesis. Wound Repair Regen 2003;11: 275-284
Hair Follicle Cell Cultures
111. Harmon CS, Nevins TD, Ducote J, Lutz D. Bisindolylmaleimide protein-kinase-C inhibitors delay the decline in DNA synthesis in mouse hair follicle organ cultures. Skin Pharmacol. 1997; 10: 71-8.
112. Imcke E, Mayer-da-Silva A, Detmar M, Tiel H, Stadler R, Orfanos CE.Growth of human hair follicle keratinocytes in vitro. Ultrastructural features of a new model. J Am Acad Dermatol. 1987; 17 : 779-86.
113. Kiesewetter F, Arai A, Schell H.Sex hormones and antiandrogens influence in vitro growth of dermal papilla cells and outer root sheath keratinocytes of human hair follicles. J Invest Dermatol. 1993; 101: 98-105.
114. Blume U, Schon MP, Zouboulis CC, Detmar M, Orfanos CE. Vellus hair follicle-derived keratinocyte culture: a new experimental model in human hair research. Skin Pharmacol.1994; 7: 27-32.
115. Limat A, French LE, Blal L, Saurat JH, Hunziker T, Salomon D. Organotypic cultures of autologous hair follicle keratinocytes for the treatment of recurrent leg ulcers. J Am Acad Dermatol. 2003; 48: 207-14.
116. Tobin DJ, Paus R. Graying: gerontobiology of the hair follicle pigmentary unit. Exp Gerontol. 2001; 36: 29-54.
117. Havlickova B, Biro T, Mescalchin A, Arenberger P, Paus R. Towards optimization of an organotypic assay system that imitates human hair follicle-like epithelial-mesenchymal interactions. Br J Dermatol. 2004 ; 151: 753-65.
118. Jahoda C, Oliver RF. The growth of vibrissa dermal papilla cells in vitro. Br J Dermatol. 1981; 105:623-7.
119. Jahoda CA, Horne KA, Oliver RF. Induction of hair growth by implantation of cultured dermal papilla cells. Nature. 1984; 311: 560-2.
120. Jahoda CA, Reynolds AJ, Oliver RF. Induction of hair growth in ear wounds by cultured dermal papilla cells. J Invest Dermatol. 1993; 101: 584-90.
121. Jahoda CA, Reynolds AJ, Chaponnier C, Forester JC, Gabbiani G.Smooth muscle alpha-actin is a marker for hair follicle dermis in vivo and in vitro. J Cell Sci. 199l; 99:627-36.
122. Almon d-Roesler B, Schon M, Schon MP, Blume-Peytavi U, Sommer C, Loster K, Orfanos CE. Cultured dermal papilla cells of the rat vibrissa follicle. Proliferative activity, adhesion properties and reorganization of the extracellular matrix in vitro. Arch Dermatol Res. 1997; 289:698-704.
123. Almeida Jr. H, Müller F, Zigrino P, Korge B, Krieg T, Mauch C . Expression and activation of matrix metalloproteinases in cultured human dermal papilla cells. Ann Dermatol Venereol 2002; 129:1S515.
124. Thornton MJ, Hibberts NA, Street T, Brinklow BR, Loudon AS, Randall VA.Androgen receptors are only present in mesenchyme-derived dermal papilla cells of red deer (Cervus elaphus) neck follicles when raised androgens induce a mane in the breeding season. J Endocrinol. 2000; 168:401-8.
125. Randall VA, Hibberts NA, Hamada K.A comparison of the culture and growth of dermal papilla cells from hair follicles from non-balding and balding (androgenetic alopecia) scalp. Br J Dermatol. 1996; 134: 437-44.
126. Han JH, Kwon OS, Chung JH, Cho KH, Eun HC, Kim KH. Effect of minoxidil on proliferation and apoptosis in dermal papilla cells of human hair follicle. J Dermatol Sci. 2004; 34:91-8.
127. Tang L, Bernardo O, Bolduc C, Lui H, Madani S, Shapiro J. The expression of insulin-like growth factor 1 in follicular dermal papillae correlates with therapeutic efficacy of finasteride in androgenetic alopecia. J Am Acad Dermatol. 2003; 49:229-33.
Other Cutaneous Cell Culture
128. Fradette J, Larouche D, Fugere C, Guignard R, Beauparlant A, Couture V, Caouette-Laberge L, Roy A, Germain L. Normal human Merkel cells are present in epidermal cell populations isolated and cultured from glabrous and hairy skin sites. J Invest Dermatol. 2003; 120: 313-317.
129. Fukuda J. A pure, monolayer culture of Merkel cells from sinus hair follicles of the rat. Neurosci Lett. 1996; 216:73-76.
130. Krasagakis K, Almond-Roesler B, Geilen C, Fimmel S, Krengel S, Chatzaki E, Gravanis A, Orfanos CE. Growth and characterization of a cell line from a human primary neuroendocrine carcinoma of the skin (Merkel cell carcinoma)in culture and as xenograft. J Cell Physiol. 2001; 187:386-391.
131. Lee CM, Carpenter F, Coaker T, Kealey T. The primary culture of epithelia from the secretory coil and collecting duct of normal human and cystic fibrotic eccrine sweat glands. J Cell Sci 1986; 83:103-18.
132. Uchida N, Oura H, Nakanishi H, Urano Y, Arase S. Dispersed cell culture of human sweat duct cells under serum-free conditions. J Dermatol 1993; 20: 684-690.
133. Schon M, Benwood J, O’Connell-Willstaedt T, Rheainwald JG. Human sweat gland myoepithelial cells express a unique set of cytokeratins and reveal the potential for alternative epithelial and mesenchymal differentation states in culture. J Cell Sci 1999; 112:1925-36.
134. Pedersen PS. Human sweat duct cells in primary culture. Basic bioelectric properties of cultures derived from normals and patients with cystic fibrosis. In Vitro Cell Dev Biol 1989; 25:342-52.
135. Shikiji T, Minami M, Inoue T, Hirose K, Oura H, Arase S. Keratinocytes can differentiate into eccrine sweat ducts in vitro: involvement of epidermal growth factor and fetal bovine serum. J Dermatol Sci. 2003; 33: 141-50.
© 2004 Egyptian Dermatology
Online Journal
|












